Monocytopenia, monocyte morphological anomalies and hyperinflammation characterise severe COVID-19 in type 2 diabetes
The year 2020 has been marred by the emergence of SARS-CoV-2 affecting over 4.5 million people globally. The resulting coronavirus disease COVID-19 has placed unprecedented strain on health services and claimed over 300 thousand lives.
Early in the COVID-19 pandemic, type 2 diabetes (T2D) was marked as a risk factor for severe disease and mortality (diabetic patients accounted for approximately 10% of COVID-19 deaths). Urgent mobilisation of diabetologists and diabetes researchers is crucial to 1) elucidate criteria that confer risk of disease and mortality in patients with diabetes, and 2) define the most suitable care.
Inflammation is central to the aetiology of both conditions where variations in immune responses can mitigate or aggravate disease course. Identifying at-risk groups based on immunoinflammatory signatures is valuable in directing personalised care and developing potential targets for precision therapy.
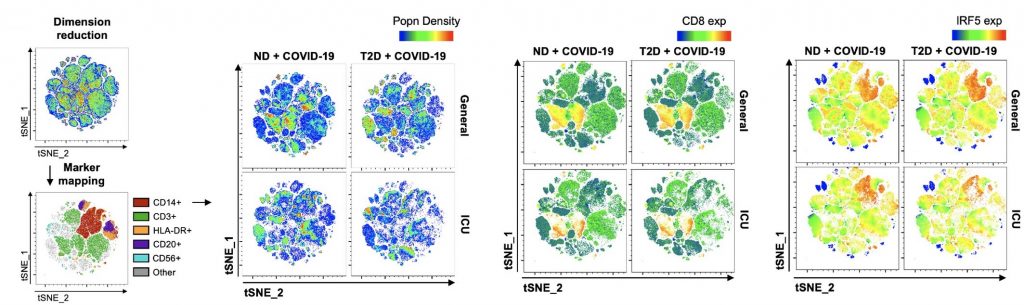
In the present study, authors characterised immunophenotypic variation associated with COVID-19 severity in patients with T2D. Broad-spectrum immunophenotyping quantified 15 leucocyte populations in peripheral circulation from a cohort of 45 hospitalised COVID-19 patients with and without T2D.
T2D patients with COVID-19 were characterised by monocytopenia,specific to quiescent monocytes. Monocyte loss was accompanied by morphological alterations and a hyperinflammatory expression profile consistent with the type 1 interferon response. These phenomena, alongside a marked loss of CD8+T cells in peripheral circulation, characterise severe COVID-19 in pre-existing T2D.
These findings allow precise identification of T2D patients with severe COVID-19 as well as provide evidence that the type 1 interferon pathway may be an actionable therapeutic target for future studies.
To read the full paper, head to the following link: